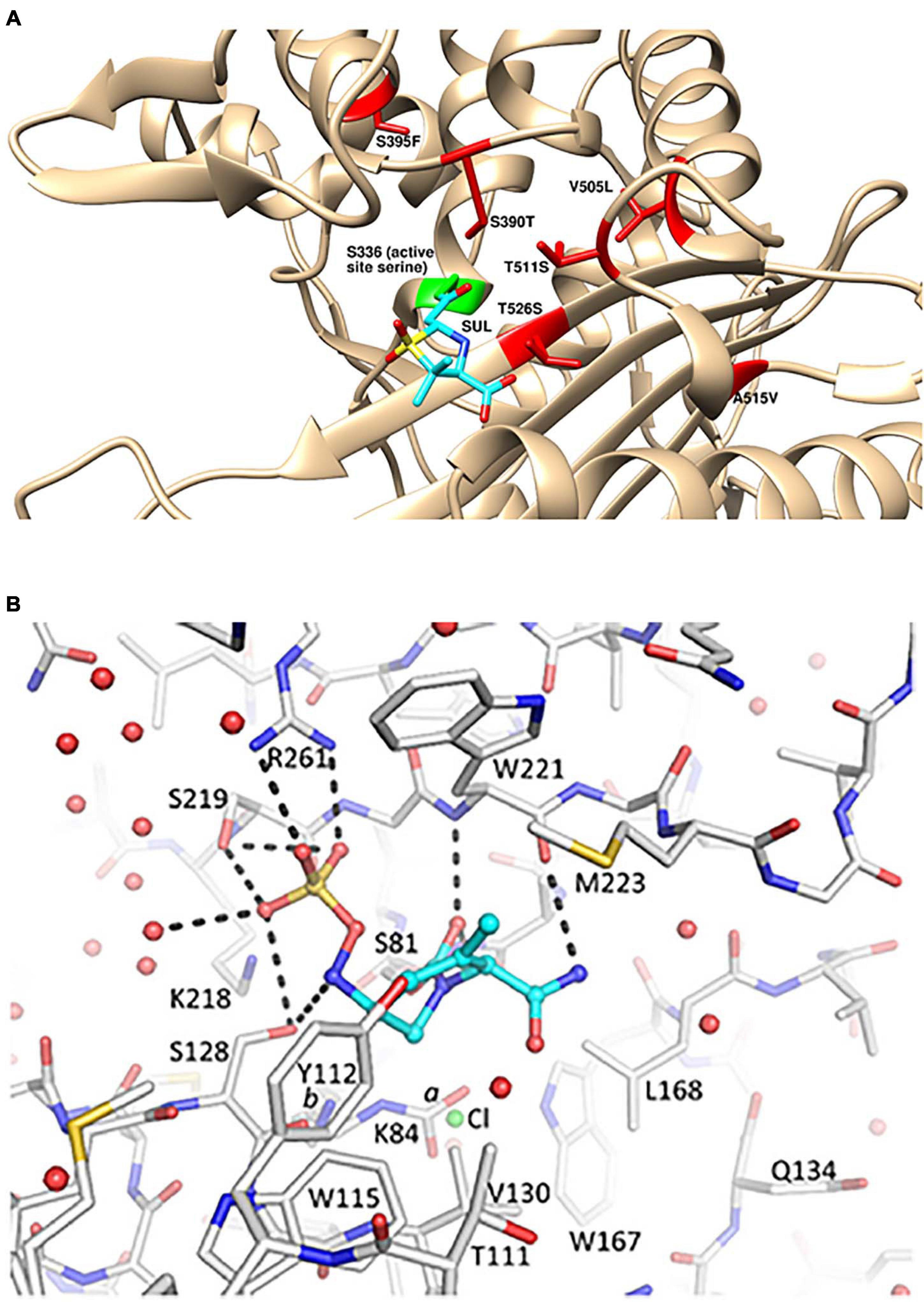
Furthermore, we explore temocillin's mechanism of β-lactamase inhibition with a high-resolution complex structure of CTX-M-14 class A serine β-lactamase. This hypothesis is supported by the observation that the acyl-enzyme complex of temocillin has reduced thermal stability compared with ticarcillin. Most notably, the 6-α-methoxy group disrupts a high-quality hydrogen bond with a conserved residue important for ligand binding while also being inserted into a crowded active site, possibly destabilizing the active site and enabling water molecule from bulk solvent to access and cleave the acyl-enzyme bond. Complex crystal structures with PBP3 reveal similar binding modes of the two drugs but with important differences. aeruginosa We show that the 6-α-methoxy group perturbs the stability of the PBP3 acyl-enzyme, which manifests in an elevated off-rate constant ( k off ) in biochemical assays comparing temocillin with ticarcillin. Here, we analyze the reaction kinetics, protein stability, and binding conformations of temocillin and ticarcillin with penicillin-binding protein 3 (PBP3), an essential PBP in P. Here, we investigated genome-wide climate adaptation in giant sequoia and coast redwood, two iconic.

Rapid changes in the climate pose significant challenges to the survival of natural populations, and the genomic basis of long-generation plant species is still poorly understood. aeruginosa have remained relatively unexplored. Dissecting the genomic basis of local adaptation is a major goal in evolutionary biology and conservation science. The origins of temocillin's inferior antibacterial properties against P. The α-methoxy modification confers resistance to serine β-lactamases, yet temocillin is ineffective against P. Temocillin is the 6-α-methoxy analogue of ticarcillin, a carboxypenicillin with well-characterized antipseudomonal properties. It has the information ensuring that PBP3 fulfils its cell septation activity within the fully complemented divisome.The prevalence of multidrug-resistant Pseudomonas aeruginosa has led to the reexamination of older "forgotten" drugs, such as temocillin, for their ability to combat resistant microbes. The E206-V217 peptide segment is exposed at the surface of the non-catalytic module.

lactams block the activity of the transpeptidases and carboxypeptidases by acylating the active site serine residue. They contain the information ensuring that PBP3 folds correctly and the acyl transferase catalytic centre adopts the active configuration. PBP3 and other penicillin-binding proteins are inhibited by -lactam antibiotics which act as suicide substrates 2, 3 by mimicking the D-alanyl-D-alanine stem peptide of the peptidoglycan precursors. Motif 1 and the I74-L82 overlapping peptide segment, motif 2 and the H160-G172 overlapping peptide segment, and the G188-D197 motif 3 are located at or close to the intermodule junction. The membrane anchor and the G40-S70 sequence (containing the G57-Q66 peptide segment) upstream from the non-catalytic module have the information ensuring that PBP3 undergoes proper insertion within the divisome at the cell septation site. Based on these data and the results of site-directed mutagenesis of motifs 1-3 and of peptide segments of high amphiphilicity (identified from hydrophobic moment plots), the M1-I236 polypeptide of PBP3 appears to be precisely designed to work in the way proposed. The fold adopted by the R71-V577 polypeptide of PBP3 has been modelled by reference to the corresponding R76-S634 polypeptide of the class B Streptococcus pneumoniae PBP2x. It has been proposed that during cell septation the peptidoglycan crosslinking activity of the acyl transferase module of PBP3 is regulated by the associated M1-I236 polypeptide itself in interaction with other components of the divisome. Previously, b-lactamase activity of a PBP subjected to point mutations or protein engineering based on the structural comparison of these two groups of enzymes 15 17 has been.

The class B M1-V577 penicillin-binding protein (PBP) 3 of Escherichia coli consists of a M1-元9 membrane anchor (bearing a cytosolic tail) that is linked via a G40-S70 intervening peptide to an R71-I236 non-catalytic module (containing the conserved motifs 1-3) itself linked via motif 4 to a D237-V577 catalytic module (containing the conserved motifs 5-7 of the penicilloyl serine transferases superfamily). Sequence, structural and catalytic analyses suggest that serine b-lactamases evolved from the more ancient PBPs 811 and several studies have shown that some PBPs display b-lactamase activity 1215.
